Trusted ERP Solutions Provider To Small, Medium and Large Enterprises


I hope you find this blog post insightful. If you’d like our team to handle technology in your business and bring the best ROI, click here.
ERP for Medical Device Manufacturers: Ensuring Compliance & Quality Control
Why Medical Device Manufacturing Needs ERP
The medical device industry is one of the most regulated sectors in the world. Whether it’s surgical tools, diagnostic equipment, or implants, manufacturers must ensure strict compliance, flawless quality, and traceability of every component.
Managing this with spreadsheets or disconnected systems often leads to errors, delays, and regulatory risks. This is why many businesses rely on ERP for medical device manufacturers. An ERP system integrates production, inventory, compliance, quality checks, and finances into a single, unified platform.
In this blog, we’ll explore how ERP helps medical device manufacturers stay compliant, improve quality control, and streamline operations, making it the perfect ERP for manufacturing companies in the healthcare sector.
Challenges in Medical Device Manufacturing
Manufacturing medical devices comes with unique hurdles:
- Strict regulatory compliance: adhering to FDA, ISO 13485, and CE standards.
- High-quality expectations: Ensuring zero-defect products for patient safety.
- Traceability requirements: Tracking components from suppliers to finished devices.
- Complex supply chains: Coordinating raw materials and suppliers worldwide.
- Documentation & audits: Maintaining detailed records for regulators.
Without ERP, managing these processes becomes time-consuming and risky.
How ERP Helps Medical Device Manufacturers
Using ERP for medical device manufacturers provides solutions tailored to the industry’s needs:
- Compliance Management: Automates documentation and ensures adherence to regulations like the FDA and ISO.
- Quality Control: Monitors every production stage with real-time checks and approvals.
- Traceability: Tracks every component and batch number, ensuring complete visibility.
- Supply Chain Efficiency: Coordinates global suppliers and ensures timely raw material availability.
- Document Management: Stores digital records, making audits easier.
- Cost Management:Tracks expenses, reduces waste, and improves profitability.
Key Features of ERP for Medical Device Manufacturing
1. Regulatory Compliance Tools
ERP ensures compliance with industry standards by automating reports, validation processes, and audit trails.
2. End-to-End Traceability
Every part and batch can be traced back to its source, ensuring accountability and safety.
3. Quality Management System (QMS)
ERP integrates QMS, ensuring continuous product quality and reducing defects.
4. Inventory & Supply Chain Management
Helps manage materials efficiently, preventing shortages and avoiding overstocking.
5. Document & Audit Management
Centralized documentation ensures easy access during audits and regulatory inspections.
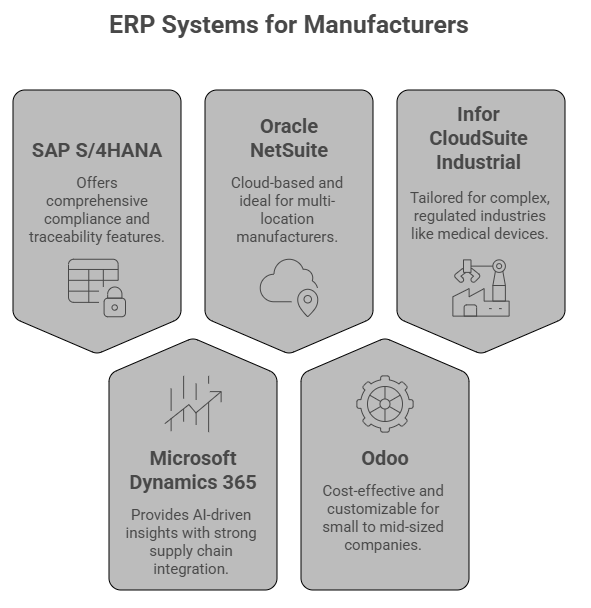
Top ERP Systems for Medical Device Manufacturers in 2025
- SAP S/4HANA: Comprehensive compliance and traceability features.
- Microsoft Dynamics 365: AI-driven insights with strong supply chain integration.
- Oracle NetSuite: Cloud-based and ideal for multi-location manufacturers.
- Odoo: Cost-effective and customizable for small to mid-sized companies.
- Infor CloudSuite Industrial: Tailored for complex, regulated industries like medical devices.
Best Practices for Implementing ERP in Medical Device Manufacturing
- Focus on compliance modules first – Prioritize features for FDA, ISO, and CE compliance.
- Ensure full traceability: Map out the supply chain and production tracking.
- Train your team: Regulatory requirements mandate that employees use ERP accurately.
- Work with industry experts: Choose ERP vendors experienced in medical device compliance.
- Test before full rollout: Pilot implementation reduces risks and ensures smooth adoption.
Conclusion
Medical device manufacturing demands precision, compliance, and quality. By adopting an ERP for medical device manufacturers, companies gain complete visibility, reduce regulatory risks, and improve product quality. The right ERP for manufacturing companies in this sector doesn’t just optimize operations; it safeguards lives by ensuring safe and reliable medical products.
ERP is crucial because it integrates compliance, production, quality, and traceability into one system. Medical device makers face strict regulations, and ERP ensures every step meets FDA and ISO standards. It reduces errors, saves time, and helps deliver safe, reliable products with full accountability and documentation.
The best ERP depends on company size and needs. SAP S/4HANA and Oracle NetSuite are great for large enterprises requiring advanced compliance tools. Microsoft Dynamics 365 offers strong supply chain management, while Odoo is affordable and customizable, making it a smart choice for small to mid-sized medical device manufacturers.
ERP simplifies compliance by automating documentation, creating audit trails, and generating regulatory reports. It ensures manufacturers follow FDA, ISO 13485, and CE requirements. With built-in validation and traceability features, ERP reduces compliance risks, keeps records accessible, and makes passing audits smoother while maintaining high product quality and safety standards.
Yes, small companies don’t need to worry about high costs. Cloud-based ERPs like Odoo and Zoho provide affordable, scalable solutions. They let smaller manufacturers start with core modules like inventory and compliance, then expand as they grow. This makes ERP accessible even for startups in the medical device industry.
Absolutely. ERP integrates quality control at every production stage, from raw materials to final assembly. It ensures defects are detected early, tracks compliance with standards, and maintains consistent product quality. This reduces recalls, improves patient safety, and builds trust with regulators and customers in the medical device sector.

Saeeda Riaz
Partner at AGN IT Services LLC | Strategic IT Partner of 10+ Enterprises
Saeeda Riaz is a seasoned IT consultant with over 23 years of experience in delivering innovative technology solutions to businesses across various industries. As the driving force behind a leading IT solutions company, she specializes in digital transformation, business automation, and software development. Her strategic vision and hands-on expertise help organizations streamline operations, enhance efficiency, and stay ahead in the digital landscape.
Connect on LinkedInContact us for IT Solutions
